OXIME - CARBOXYLIC ACID SYSTEM, ANOTHER SUPRAMOLECULAR SYNTHON
Maciej Kubicki and Teresa Borowiak
Laboratory of Crystallography, Department of Chemistry, Adam Mickiewicz University, Grunwaldzka 6, 60-780 Poznan, Poland; e-mail: mkubicki@main.amu.edu.pl.
Keywords: hydrogen bonds, supramolecular
synthons, oximes, bornane derivatives.
It is known that oximes generally tend to form six membered intermolecular oximetooxime dimers, hydrogen-bonded through O-H...N bonds [1]. This picture can be complicated by the presence of other functional groups, having a possibility of donating or accepting hydrogen bonds. In particular, molecules containing both oxime and carboxyl groups almost always make mixed oxime-carbonyl hydrogen bonds, which close seven-membered rings.
It was also suggested [2] that the p-electron resonance of the oxime and carboxyl fragments has a significant influence on the O-H...O and O-H...N hydrogen bond lengths (and, consequently, their strengths), causing the shortening of the former and the lengthening of the latter in comparison with the compounds without such resonance.

In order to check these tendencies, we have studied the
crystal structures of two bornane oximes:
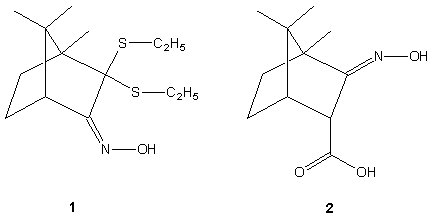
2,2'-diethylthiobornane-3-oxime (1) and bornane-2-oxime-3-endo-carboxylic
acid (2). As expected, in crystal structures of both
compounds the molecular packing is determined mainly by
intermolecular hydrogen bonds. In 1, the oxime-to-oxime
hydrogen bonds between two symmetry independent molecules close
six-membered ring. In 2 there are only oxime-to-carboxyl
hydrogen bonds. The robustness of oxime-carboxyl motif allows to
regard it as the supramolecular synthon (in a sense given by
Desiraju [3]), which could be helpful in joining the molecules
into polar chains. This is further confirmed by the formation of
1:1 complexes between oximes and carboxylic acids [4]. The
details of these structures together with the database search
results will be also given.