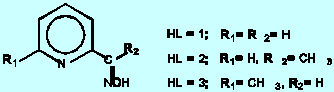
CRYSTAL STRUCTURE OF A NONAMERIC NICKEL(II) METALLACROWN COMPLEX, C70H88Cl2N20Ni9O30.10H2O
Aarne Pajunen, Ilpo Mutikainen, Marjatta Orama and Heikki Saarinen
Division of Inorganic
Chemistry, Department of Chemistry, University of Helsinki, Box
55, FIN-00014 UNIVERSITY OF HELSINKI, Finland, E-mail:
aarne.pajunen@helsinki.fi
In the course of our structural and equilibrium studies on transition metal complexes formed by different oxime ligands, we have shown that pyridine-2-carboxaldehyde oxime compounds of the type
readily form polynuclear complexes which are potential model compounds in the study of redox and magnetic-exchange interactions of metal ions [1]. The best known examples are the trinuclear copper(II) complexes characterized by the presence of a highly stable trinuclear Cu3OH central core both in the solid state as well as in aqueous solution [2].
With nickel(II), on the other hand, most of the complexes formed by ligands HL =1 and HL = 2 have been found to belong to the mononuclear series Ni(HL)n2+ (n = 1,2,3) or are oximato members of the series derived from the stepwise dissociation of the oxime protons from the parent bis and tris complexes [3]. The 6-methyl group on the pyridine ring, however, seems to have a crucial effect on the polymerization reactions of nickel(II) and binuclear and trinuclear complexes of the composition Ni2(HL)3H-3+ and Ni3(HL)3H-5+ are readily formed as major species in aqueous solution when HL = 3 [4].
To facilitate interpretation of the coordination models in these polynuclear structures we have now isolated one of the complexes formed between nickel(II) and HL = 3 under aqueous condition where polymerization is known to occur in the solid state and analyzed it by X-ray diffraction methods.
The compound crystallizes in the space group P(-1) with a = 15.920(7), b = 16.673(8), c = 20.583(10) Å, a = 77.80(6), b = 81.00(6) , g = 72.60(4)o, Z = 2. Full-matrix least-squares refinement of 12584 reflections collected by means of a Rigaku AFC7S diffractometer yielded R = 0.1084.
The crystalline species isolated as perchlorate from solution,
where the trinuclear species -5,3,3 predominates proved to have a
very complicated [Ni9L10(HO)6(H2O)6]2+
complex moiety, which in our formulation corresponds to the
notation of -16,9,10. This metallacrown [5] structure contains an
octahedral NiO6 central core and four different
octahedral NiO4N2 and NiO2N4
environments. All the oxime groups are deprotonated and the nine
nickel atoms are linked together via several bi- or trifurcated
oximato and hydroxo bridges.
1. Orama, M. and Saarinen, H. Acta Chem
Scand., Ser A 50 (1996) 1087-1091.
2. Orama, M. Saarinen, H. and Korvenranta, J. J. Coord. Chem.
22 (1990) 183-190
3. Orama, M., Saarinen, H. and Korvenranta, J., Acta Chem.
Scand., Ser A 43 (1989) 407-412.
4. Saarinen, H. and Orama, M. Acta Chem Scand.
52 (1998) To be published.
5. Pecoraro, V. L., Stemmler, A. J., Gibney, B. R., Bodwin, J.
J., Wang, H., Kampf, J. W. and Barwinski, A. Progress in
Inorganic Chemistry, Vol 45 (1997) 83-177.