MOLECULAR STRUCTURE AND CONFORMATION OF NOVEL TETRADENTATE OPEN-CHAIN OXIME LIGANDS
I.O.Fritsky1, V.A.Kalibabchuk2, E.V.Prisyazhnaya1, T.Sliva1, T.Glowiak3, H.Kozlowski3
1Department of Chemistry,
Shevchenko University, Kiev 252033, Ukraine
2Department
of General Chemistry, National Medical University, Kiev 252004,
Ukraine
3Faculty
of Chemistry, University of Wroclaw, 50-383 Wroclaw, Poland

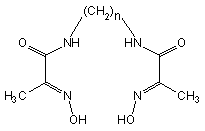
Three new open chain
oxime-and-amide ligands, derivatives of alifatic diamines and
2-hydroxyiminopropanic acid:
N,N'-bis(2-hydroxyimino-propionyl)-1,2-diaminoethane (1, n
= 2); -1,3-diamino-propane (2, n = 3) and
1,4-diaminobuthane (3, n = 4) appeared to be a very
efficient chelating agent for the Ni2+ and Cu2+
ions exhibiting both open-chain and pseudomacrocyclic
co-ordination modes in the complexes [1]. As the number of the
methylene groups and conformational behaviour of the ligands can
impact critically on the donor ability, we have studied their
crystal and molecular stucture.
In 1-3
the oxime groups are in trans-position in respect to the
amide carbonyl, the amide bonds indicate trans-configuration.
The molecule 1 (Fig.1) is centrosymmetric and
consists of two planar parallel fragments linked by the
C(4)-C(4a) bond that lies nearly perpendicular to the fragments
(the torsion angle C(3)N(2)C(4)C(4a) = 85.4(2)o) thus
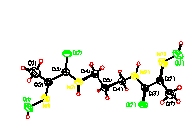
forming the „double desk" conformation. The molecule 2 (Fig.2) indicates
elongated conformation and consists of two nearly planar
fragments CH3-C(=NOH)-C(O)NH-CH2- linked by
the methylene group. The conformationally flexible fragment -(CH2)3-
lies nearly in the plane of the amide group O(2)C(3)N(2)C(4) (the
torsion angles C(3)N(2)C(4)C(5) = -176.2(2)o and
N(2)C(4)C(5)C(4') = -171.9(2)o). The molecule 3 (Fig.3)
possesses a planar „zig-zag"-like conformation with the
torsion angles deviating no more than 8o from the
values of 0o or 1800. The CH3-C(=NOH)-C(O)NH-
fragments in 3 are slightly deviated from planarity
because of turnover of the oxime and amide groups along C(2)-C(3)
and C(2')-C(3') bonds (the dihedral angles are 4.6 and 6.7o,
respectively). The conformation of both 1 and 3 is
different from that of 2 exhibiting rather folded
conformation with the dihedral angle between the planes of the
fragments = 96.2o.
1. A.M. Duda , A. Karaczyn , H. Kozlowski, I.O. Fritsky, T. Glowiak , E.V. Prisyazhnaya, T.Yu. Sliva, J. Swiatek-Kozlowska, J.Chem.Soc., Dalton Trans., 1997, 3853.
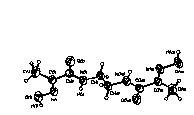 |
 |
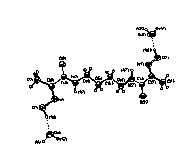 |
| Fig.1 | Fig.2 | Fig.3 |