STRUCTURE OF NEUTRAL SIX-COORDINATED ORGANIC DERIVATIVES OF SB(V)
G.K. Fukin, L.N. Zakharov, G.A. Domrachev
G.A. Razuvaev
Institute of Organometallic Chemistry, Russian Academy of
Sciences, Nizhny Novgorod, 603600, Russia
E-mail:gera@imoc.sinn.ru
Keywords: X-ray analysis,
molecular and crystal structure, donor-acceptor complexes,
quantum - chemical calculations
Neutral organic donor-acceptor complexes of a type R5Sb... L are known essentially less, than their halogen derived analogs. On a basis of the Cambridge bank of structural data [1] to the present time the structures of about 1500 compounds of three and five-valent antimony, accordingly ~740 and ~760 structures are defined. However the majority of described Sb(V) compound structures are cationic-anionic pairs (~600 structures), from which about 500 structures include fragments of a type [SbHal6]-1. Besides the overwhelming amount of neutral structures of investigated five and six-coordinated compounds of Sb(V) are halogen derived complexes, which structure is largely determined by an electrostatic situation in a coordination sphere of atoms Sb(V).
In this work the systematic X-ray researches of crystalline products of one-step oxidation reactions of Ph3Sb and Me3Sb by hydroperoxides at the presence of various organic molecules are carried out. A basic problem of this work is a definition of molecular and crystalline structures of obtained compounds, study of coordination possibilities of valent saturated Sb(V) atom, "dynamics" of a structure modification of neutral organic Sb(V) compounds depending on the nature of ligands and research of influence of the steric and electrostatic factors for forming the donor-acceptor bond.

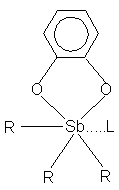
As a result of systematic X-ray researches the molecular and crystalline structure of five new neutral six-coordinated Sb(V) derivatives: Ph3(C6H4O2)Sb... L, L = OSMe2 (1), ONC5H5 (2), OPPh3 (3) [2]; Me3(C6H4O2)Sb... L, L = ONC5H5 (4), NC5H5 (5) was defined.
Besides the data of the X-ray analysis, the complexes 4 and 5 were used as initial for quantum - chemical calculations. Their purpose was a definition of the nature of donor-acceptor bond in these complexes.
In a result of our research there was shown, that 1) the distances Sb... L are varied in a wide range of values (2.266(5) - 2.595(3) A), 2) the distances Sb... L in neutral six-coordinated derivatives of Sb(V) are longer, than in similar halogen - derived complexes, 3) the distances Sb... L depend on donor ability L and R, and also on the repulsion energy between acceptor and donor molecules, 4) the nature of Sb... L bond varies from hardly polar covalent up to ionic.