NEW STRUCTURE TYPE IN THE BA-IN-CU-SYSTEM
Gerlinde Frenzen
FB Chemie-Biologie, Universität Kassel,
D-34109 Kassel
email: frenzen@fkc-ws1.chemie.uni-kassel.de
Keywords: Intermetallic Compound,
Crystal Structure, Electronic Structure
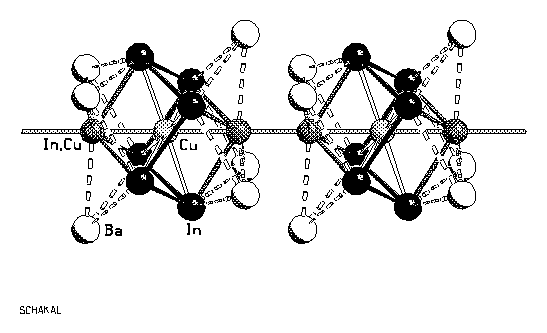
Investigation of the system Ba-In-Cu revealed
the new compound Ba6In7Cu2 .
Crystals of the compound are prepared by reaction of the elements
in sealed steel tubes at 1000°C and subsequent annealing at
600°C. Ba6In7Cu2 crystallizes
in the trigonal space group
R with the cell dimensions: a = 1491.6(4), c = 743.2(2) pm, Z =
3. The structure consists of linear chains of In7Cu2
cubes, connected by direct bonds between vertices. Cu occupies
the centers of the cubes, the occupation of the bridging vertices
is disordered with 50% copper and 50 % indium. The bond lengths
for the indium atoms are: 274.1(1) pm to the central copper atom,
305.3(1) pm to the bridging vertex and 2 x 314.2(1) pm to the
other indium vertices. The cube is slightly distorted: distance
copper - bridging vertex: 249.9(2) pm (2x), Cu - In: 274.1(1) pm
(6x). The distance between the bridging vertices is
243.5(3) pm, they are surrounded by three Ba atoms in the
remarkably short distance of 338.4(1) pm. The chemical bonding is
discussed with semiempirical band structure and COOP
calculations.
