COMPARISON OF STEREOCHEMICAL CHARACTERISTICS OF DONOR-ACCEPTOR INTERACTIONS IN DECABORANE(12) COMPLEXES WITH SUBSTITUTED PYRIDINES
T.M. Polyanskaya
Institute of the Inorganc Chemistry Siberian Branch RAS, 630090 Novosibirsk, Russia
Decaborane(12) complexes, 6,9-B10H12L2 (L is Py and its derivatives), are of interest as a new class of luminophores. A quantum-chemical calculation (MNDO) shows that the orientation of heterocyclic L affects the participation of MO's in the formation of the donor-acceptor B-N bond and the transfer charge peculiarities 1. The luminescent properties are due to the process of the back donation from the acceptor to the donor, |B10H12|->L.
Using X-ray diffraction data we
have performed a comparative analysis the B-N bond length,
orientational characteristics and relative luminescence quantum
yield (I) (data from 2) for 11 decaborane complexes with L=Py;
2-Pic (picoline);3-Pic; 4-Pic; 2,4-Lut (lutidine); 2,5-Lut;
2-Me-5-Et-Py; 3,4-Lut; 3-CN-Py;.3-Br-Py (see Table and Fig.).
L |
Symmetry of the complex | pKa | d, A | Twist type |
Angle w (o) | Angle j1 (o) | Angle j2 (o) |
I |
| 3-CN-Py | N1 | 1.35 | 1.597(3) | cis | 18.7 | -45.4 | -53.0 | 0.04 |
| Py-I | C1 | 5.22 | 1.573(3) | trans | 75.8 | 52.5 | -53.9 | 0.35 |
| Py-II | C1 | 5.22 | 1.573(1) | trans | 80.8 | -38.3 | 46.1 | 0.39 |
| 3-Pic | C1 | 5.63 | 1.578(4) | trans | 74.3 | -50.5 | 56.2 | 0.38 |
| 2-Pic | C1 | 5.96 | 1.579(3) | trans | 89.5 | -45.1 | 48.3 | 0.20 |
| 4-Pic | C2 | 5.98 | 1.576(2) | trans | 89.9 | -45.6 | 45.6 | 0.66 |
| 2,5-Lut | C1 | 6.40 | 1.580(7) | cis | 15.9 | 44.4 | 51.4 | 0.18 |
| 3,4-Lut | CS | 6.46 | 1.582(4) | cis | 23.6 | 39.4 | 39.4 | 1.0 |
| 2,4-Lut | C2 | 6.63 | 1.582(2) | trans | 83.44 | 48.4 | -48.4 | 0.63 |
| 2-Me-5-Et-Py | C1 | 1.583(8) | cis | 3.2 | 45.0 | 47.7 | 0.42 | |
| 3-Br-Py | C2 | 1.574(7) | cis | 58.6 | 30.2 | 30.2 | 0.02 |
For substitutes that enhance the
donor properties of L (pKa increases), the -type B-N
bond is shorter than the covalent bond 1.59 A in BN. The sequence of its stretching, Py->monosubstituted Py->disubstituted Py, is nearly the same as
the sequence of the change in pKa. This is due to the
compromise between two rival processes, i.e., a compression
because of the back donation and a stretch because of the steric
strains. For L=3-CN-Py with electron-seeking properties, d=1.597
A exceeds the
covalent bond and virtually equal to that for L=NH3
with a preferentially s-type bond.

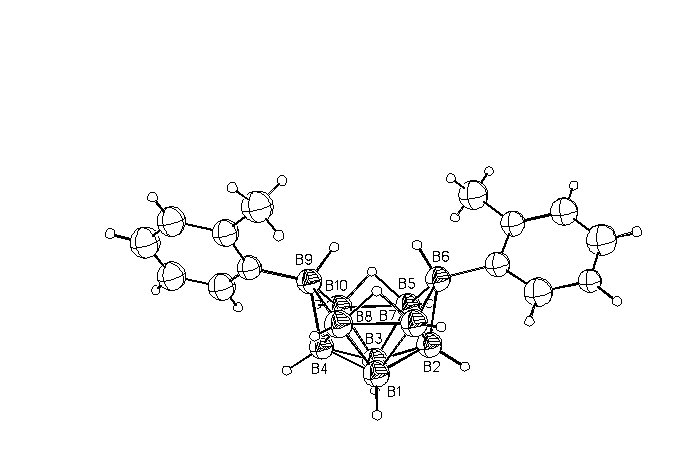
Figure. Perspective
drawing of the unit molecule 6,9-(2-Pic)2(B10H12).
For L=Py there are two modifications that differ in a mutual orientation of the Py molecule planes relative to each other (w angle) and the B(2)B(4)B(6)B(9) plane (A-plane) of the |B10H12 |cluster (j1 and j2 angles) at the same opposite-side twist relative to the latter plane (the trans-twist in Table). As seen from Table, a variation of the substitutes and their positions in the Py ring results in complexes with different symmetry. There occurs not only trans- but also cis-twist of the Py-ring planes relative to the A-plane with a different orientation of the Py rings relative to each other that varies from near parallel in 2-Me-5-Et-Py to near orthogonal in 2-Pic and 4-Pic.
Among complexes with similar substitutes, the high-symmetry molecular complexes with symmetry Cs (3,4-Lut) and C2 (4-Pic and 2,4-Lut) exhibit an enhanced luminescence quantum yield. Although the luminescence of the two latter compounds is high at the trans-twist of the Py cycles, it achieves the maximum at the cis-twist in 3,4-Lut with the same angles (j1=j2=39.4o). None of the compounds in the condensed phase has the orientation with the above angles equal to 0 that was proposed to be optimal for the efficient back donation [3].
Not only orientational effects influence the luminescence quantum yield, it decreases, perhaps, due to the p-stacking interactions of aromatic heterocyclic Py cycles of the neighboring complexes of the dipole-dipole type observed in some crystal structures (e.g., with L=Py-I, 2-Pic, 2,5-Lut).
An increase in the luminescence quantum yield is attended with a loss in the packing efficiency that is not balanced out by strengthening the donor-acceptor B-N bond.
The coplanarity of 5-membered cycles (involving B(6) and B(5) (or B(7)) atoms on an exposed surface of the nido-cluster and N, C, Hc atoms of the Py ring) and the Py rings is disturbed in complexes with good luminescence; the torsion CNB(6)B(7) angle is 24.5o in the complex with L=3,4-Lut.
1. V.V.Murachtanov,
T.M.Polyanskaya, V.V.Volkov: Zh. Stukt. Khimii, 37 (1996),
652-663.
2. E.A.Il'inchik,
V.V.Volkov, S.T.Dunaev: Zh.Strukt.Khimii, 37 (1996),
59-67.
3. B.M.Graybill,
M.F.Hawthorne:J. Amer. Chem. Soc., 83 (1961),
2673-2676.