 |
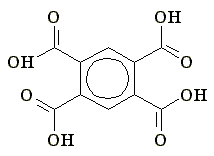 |
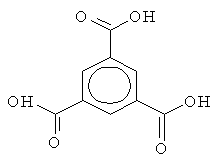
| Fig 1. 1,3,5-Benzenetricarboxylic acid. | Fig. 2. 1,2,4,5-Benzentetracarboxylic acid. |
BENZENE POLYCARBOXYLATE COMPLEXES OF SODIUM AND METHANOL CLATHRATE COMPOUNDS: THE RESULTS OF AN UNDERGRADUATE RESEARCH PROJECT
Mark R.J. Elsegood and Simon J. Richards.
Department of Chemistry, Bedson Building, The University of Newcastle-upon-Tyne, Newcastle-upon-Tyne, NE1 7RU.
Benzene polycarboxylates are of current interest for several reasons. They have wide actual and potential application as fire retardants, catalysts, and as reaction templates due to their similarities to zeolites in their solid-state packing arrangements, as well as many other uses. Their solid state architecture is dominated by hydrogen bonding, but also often exhibits cross-linked x-y sheets with p-stacking of the benzene rings. Thus there are crystal engineering aspects to this research area also.
We have been investigating alkali metal complexes of
1,3,5-benzenetricarboxylic acid (Fig. 1) and
1,2,4,5-benzenetetracarboxylic acid (Fig. 2) and have obtained
structural results for the monosodium salt of the former and the
disodium salt of the latter. As part of these studies we have
additionally solved structures for two methanol clathrate
complexes of the 1,3,5 acid, one with a methanol:acid ratio of
1:1, the other 2:1. All of these compounds have interesting
solid-state packing motifs and some raise interesting
crystallographic issues. X-ray crystallography has been
particularly important in compound characterisation in this work
as the n.m.r. spectra of these compounds are not especially
informative. The crystals tend to be unstable at ambient or
higher temperatures due primarily to solvent loss. All data sets
were measured at low temperature using a Bruker SMART CCD area
detector diffractometer.
 |
 |
| Fig 1. 1,3,5-Benzenetricarboxylic acid. | Fig. 2. 1,2,4,5-Benzentetracarboxylic acid. |