SELECTIVE INCLUSION WITH A HEXAPEDAL HOST
Susan A Bourne1, Katherine
L Gifford Nash1, Fumio Toda2
1 Department of Chemistry, University of Cape Town, Rondebosch 7701, South Africa
2 Department of Applied Chemistry,
Faculty of Engineering, Ehime University,
Matsuyama 790, Japan
Keywords: supramolecular chemistry,
hexahosts, selective inclusion
Three basic aspects of supramolecular chemistry include
molecular recognition, supramolecular reactivity and catalysis
and transport processes [1]. Molecular recognition involves
chiefly the selective complexation of the host-guest type. A host
that forms complexes of different stabilities with two similar
guests is said to show molecular recognition and such selective
complexation plays an essential role in the various aspects of
supramolecular functions. It has been observed that in
order to achieve selectivity a host molecule must be rigid so as
to allow a preorganised binding site which is complementary in
size and polarity to a particular guest [2].
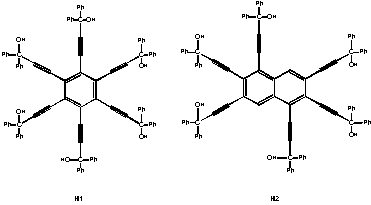
In recent years, we have studied a series of host compounds which fit the criteria above, and have, in addition, a relatively large number of hydrogen-bonding donor sites. This paper will present the results of our studies on two such host compounds: hexakis(3-hydroxy-3,3-diphenyl-2-propynyl)benzene (H1), and 1,2,3,5,6,7-hexakis(3-hydroxy-3,3-diphenyl-2-propynyl)naphthalene (H2). H1 includes a wide variety of guest molecules and exhibits specific selectivity towards carbonyl-type oxygens over etheric oxygens. H2, although similar in shape to H1, is a larger molecule and displays a preference for somewhat larger guests.

1 J-M. Lehn in Frontiers in
Supramolecular Organic Chemistry and Photochemistry, eds.
H-J. Schneider, H. Dürr,
VCH Publishers, Weinheim, New York, 1991.
2 E Weber in Inclusion compounds,
eds. J. L. Atwood, J. E. D. Davies, D. D. MacNicol,
Oxford University Press, 1991, Vol. 4.