CORRELATION OF STRUCTURAL FEATURES WITH BARRIERS TO THERMAL RACEMIZATION OF SPIROBINAPHTHOPYRANS
D. Mrvoš-Sermek,1 A. Hergold-Brundia,1 A. Nagl,2 L. Lonear-Tomaškovia,3 K. Lorenz,4 M. Mintas3 & A. Mannschreck4
1Laboratory of General and Inorganic
Chemistry, Faculty of Science, University of Zagreb, HR-10000
Zagreb, Croatia, mrvos@zagreb.zoak.pmf.hr
2Faculty of Textile Technology, University of
Zagreb, HR-10000 Zagreb, Croatia
3Department of Organic Chemistry, Faculty of
Chemical Enineering and Technology, University of Zagreb,
HR-10000 Zagreb, Croatia
4 Institut für Organische Chemie, Universität
Regensburg, D-93040 Regensburg, Germany
Keywords: chiral spirobinaphthopyrans; barriers to racemization; X-ray crystallography .
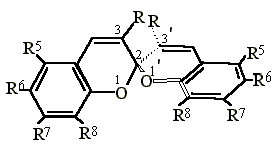
Spiropyrans have been investigated largely because of their thermo- and photochromic properties and application in display and storage of information (1). In connection with our previous studies on chiral 2H-chromenes (2,3) we have prepared the novel spirobinaphthopyrans 1-9 (Scheme) in order to separate their enantiomers and determine the unknown barriers for interconversion of their enantiomers.

| R | R5 R6 | R7 R8 | |
| 1 | CH3/CH3 | - | benzo |
| 2 | (CH2)3 | - | benzo |
| 3 | (CH2)4 | - | benzo |
| 4 | C6H5/C6H5 | - | benzo |
| 5 | CH3/CH3 | benzo | |
| 6 | (CH2)3 | benzo | |
| 7 | (CH2)4 | benzo | |
| 8 | CH2OCH2 | benzo | |
| 9 | CH2SCH2 | benzo | |
| 10 | (CH2)2 | H H | H H |
Gibbs energies of activation DG<>
for reversible clevage of the Cspiro-O bond in 1
- 9 have been found in the range 85-105 kJ/mol and are
rationalyzed by steric and electronic effects. The results of the
X-ray crystal structure analysis for the derivatives 3,
4, 6, and spirobibenzo compound 10, are in a
good agreement with the observed free enthalpies of activation.
The DG<>
values decrease with the decreasing magnitude of the bridge or
decreasing size of the substitute group on the site 3,3',
therefore DG<>
for 7>9>6, 3>2 and 4>1.
This may be explained by the destabilization of the ground state
of spiro-compounds caused by an increase of ring strain energies.
The valence angle around the spiro carbon atom [C3-C2-C3']
increases with elongation of the bridge length in 10, 6
and 3, and the longest one is in the 3,3'-
diphenylsubstituted derivative 4. This angle widening
follows the ring strain decrease in the sequence 10, 6
and 3. Thus the [C3-C2-C3'] angle in 3 118.5o is
for 13.9o and 7.3o greater than that of 10
and 6 respectively. Compared to it, values of the [O1-C2-O1']
angle in 10, 6, and 3 vary only slightly and are in
good agreement with the expected ones for the C(sp3)-carbon
atom. Furthermore, the dihedral angle between the least square
planes [C3-C2-C3'] and [O1-C2-O1'] decreases consistently with
the [C3-C2-C3'] angle broadening in the series 10, 6,
3 and 4. The mean value of the Cspiro-O
bond length in spirobibenzopyran 10 amounting 1.432 A is
shorter than that in the spirobinaphtho derivatives 4, 3
and 6 1.437-1.446 A. This bond length shortening may be
explained by the weaker conjugation of the oxygen p-lone electron pair with the p-system of the benzene ring in 10
than the one with the naphthalene moiety in 3, 4,
and 6. Therefore, the bond lengths O1-C10 and O2-C10' in 10
are longer [1.399(3) and 1.386(3) A, ] than those of 6, 3
and 4, while the Cspiro-O bonds are
shorter, that is, stronger. This bond length strengthening, may
be related with the reversible cleavage of the Cspiro-O
bond in the interconversion of enantiomers. In accord with this
we have found that the free enthalpy of activaton, DG<>
for racemization is higher for 10 than for the 4, 3
and 6.