A COMPARISON OF THE ENAMINO-CARBONYL CONJUGATION EFFICIENCY IN PYRIDONE AND DIHYDROPYRIDONE SYSTEMS
Irena Wolska, Teresa Borowiak, Artur Korzanski
Faculty of Chemistry, Adam
Mickiewicz University, Grunwaldzka 6, 60-780 Poznan, Poland
e-mail: iwolska@main.amu.edu.pl
Keywords:
pyridone, dihydropyridone, conjugation in, resonance assisted
hydrogen bonds.
Intermolecular hydrogen bonds N-H·····O display a large spectrum of N····O distances, typically from 2.70 to 3.30 Å. It has been suggested by Bertolasi et al. [1] that very short intermolecular hydrogen bonds C=O····H_N, with their N····O distances shorter than 2.90 Å, can occur only when the NH and C=O groups are connected by a p-conjugated system.
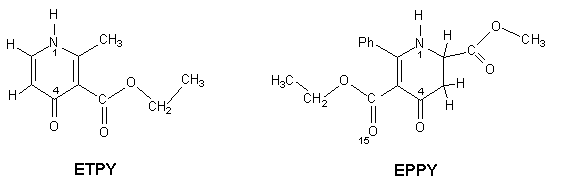
Here we present the X-ray
structures of two derivatives of 4-pyridone:
3-ethoxycarbonyl-2-methyl-4-pyridone (hereafter ETPY) and 3-ethoxycarbonyl-2-phenyl-6-methoxycarbonyl-5,6-dihydro-4-pyridone
(hereafter EPPY).

In both of them resonance paths N_C=C_C=O exist. Due to this structural similarity one can expect short intermolecular hydrogen bonds between the pyridone carbonyl and NH groups and the elongation of C=O bond caused by the resonance. This has been found in ETPY structure, the carbonyl bond lengths are 1.256(4) and 1.260(4) Åin two molecules of the asymmetric unit and the intermolecular hydrogen bond is very short with N····O distances of 2.680(4) and 2.709(4) Å. However, it is not the case of EPPY. In its crystal structure molecules are also hydrogen bonded, but to O15 of the ester group. The N····O distance is longer and equal to 2.887(2) Å and C=O15 bond lenght is 1.214(3) Å.
HOMA indices (harmonic
oscillator measure of aromaticity) [2] have been calculated for
all p-electron fragments of both molecules
and conjugation efficiency is discussed.