Fig. 1
Fig. 2
Fig. 3
POLYOXOMETALATES CONTAINING COORDINATIVELY BOUND ORGANIC LIGANDS
Neven Strukan, Marina Cindric, Maja Devic and Boris Kamenar
Laboratory of
General and Inorganic Chemistry, Chemistry Department, Faculty of
Science, University of Zagreb, Ulica kralja Zvonimira 8, HR-10000
Zagreb, Croatia
e-mail: bkamenar@public.srce.hr
Keywords: crystal structure, polyoxometalates,
molybdovanadates, oxalate, glycine.
There is a considerable interest
in the chemistry of polyoxometalates with simple organic subunits
(such as carboxyl, carbonyl, alkoxy and organonitrogen groups)
due to their importance as models for the interactions between
catalytic metal oxide surface and organic molecules. As part of
our research four novel polyoxomolybdovanadates have been
prepared and their structures determined by
X-ray diffraction. Their crystal data are:
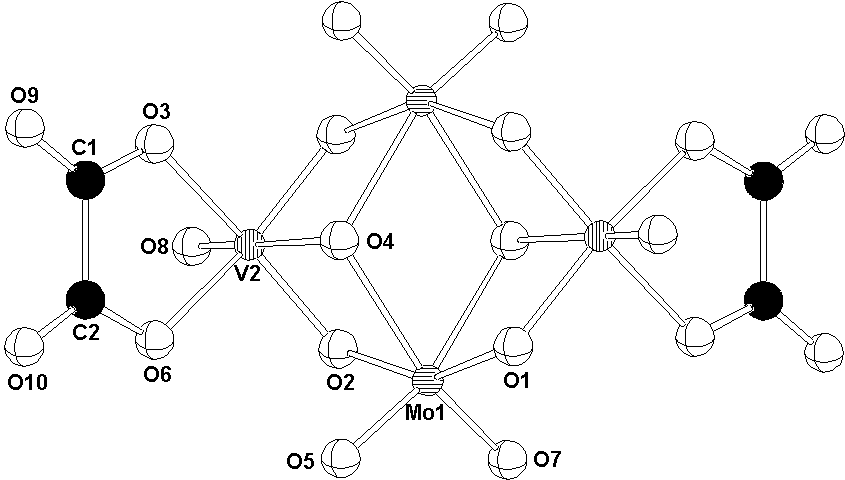
The centrosymmetric [H2Mo2V2O12(C2O4)2]4- anion of (I) is built up of two MoO6 and two VO6 edge-sharing octahedra, with bidentate oxalato ligands bonded to the vanadium atoms (Fig. 1).
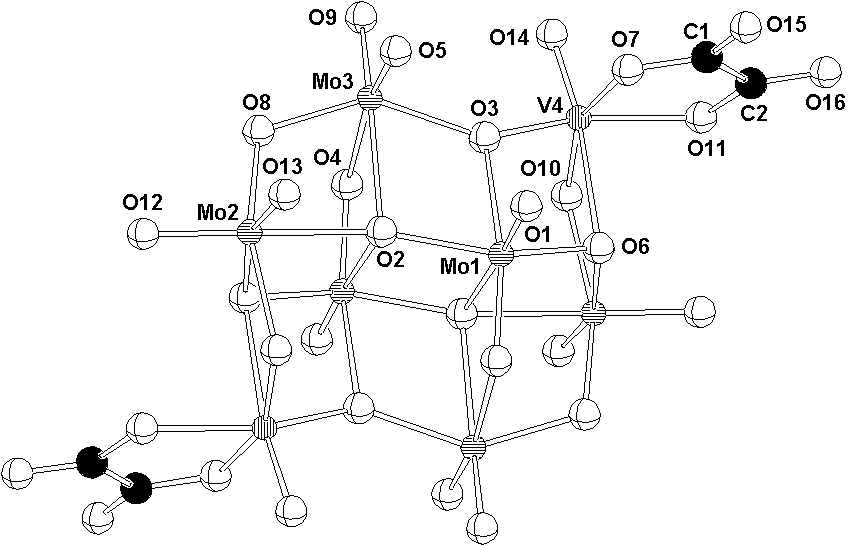
The molybdovanadate anion [Mo6V2O24(C2O4)2]6- found in (II) and (III) consists of six MoO6 and two VO6 edge-sharing octahedra, and adopts -[Mo8O26]4- type structure (Fig. 2). Bidentate oxalato ligands are also linked to the vanadium atoms.
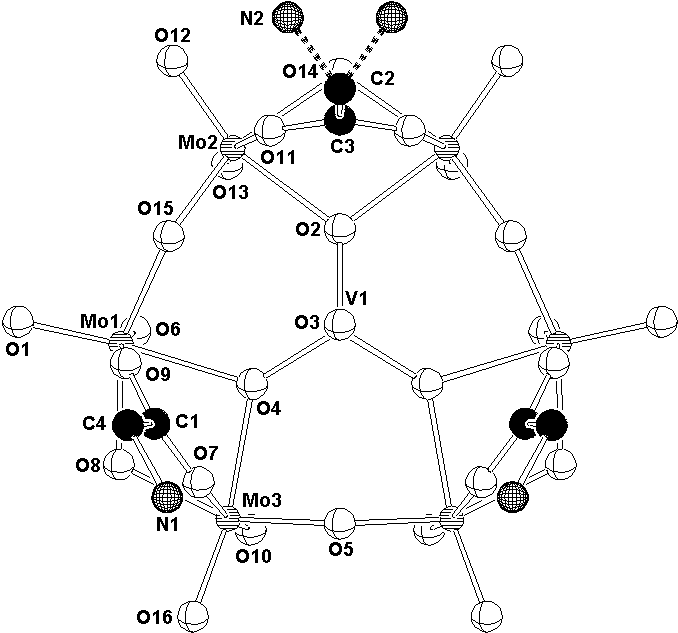
In (IV) polyanion [HMo6VO22(NH3CH2COO)3]2-
with a mirror plane symmetry is built up of six MoO6
edge-sharing octahedra connected into a ring centred by VO4
tetrahedron. The MoO6 octahedra are in pairs linked by
the bridging glycine-carboxylato ligands (Fig. 3). To keep the
compound electrically neutral one hydrogen atom must be attached
to the anion. Its position was located in a difference map.
 |
 |
 |
Fig. 1 |
Fig. 2 |
Fig. 3 |
Figure 1. Figure 2.
Figure 3.