STEREOCHEMICAL CHANGES
BY CHELATATION:
N,N'-BUTYLENEBISSALICYLALDIMINE AND ITS COPPER COMPLEX
Laboratory of General and Inorganic
Chemistry, Faculty of Science,
University of Zagreb, Ul. kralja Zvonimira, HR-10000 Zagreb,
Croatia
Keywords: symmetrically bridged Schiff
base, tetradentate ligand, copper(II) salicylaldiminate
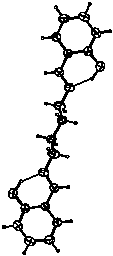
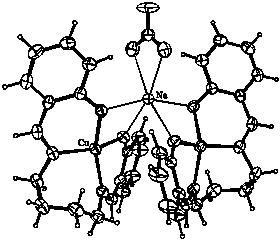
N,N'-butylenebissalicylaldimine (I) was prepared from 1,4-diaminobutane and salicylaldehide. The copper complex was prepared in a template reaction using NaOH as a deprotonation agent. The crystals of (II), obtained by slow evaporation, consist of two discrete molecules of copper complex bridged by a sodium ion.
 |
 |
| (I) | (II) |
(I) C18H20N2O2,
P 21/c, a = 15.345(1) Å, b
= 5.8799(5) Å, c = 8.9744(8) Å,
b = 90.145(8) o, V = 809.72(8) Å3, Z
= 2, Dc = 1.216 gcm-3, l(MoKa)
= 0.71073 Å,
R = 0.051 and wR = 0.089 for 1179 independent
reflections with Inet >= 2s Inet.
(II) C36H36N5NaCu2O7,
P bcn, a = 16.908(5) Å, b =
11.409(4) Å, c = 18.172(5) Å,
V = 3505.4(19) Å3, Z = 4, Dc
= 1.517 gcm-3, l(MoKa) = 0.71073 Å, R =
0.039 and wR = 0.058 for 1975 reflections with Inet
>= 2s Inet.
The main feature of (I) is the existence of an
O-H···N intramolecular chelate ring hydrogen bond. It
was assumed that the length of the bridging chain in (I)
could introduce certain strain in the molecule of its CuII
complex thus influencing to the geometry of copper atom
coordination sphere. The sodium ion bridges two metal complex
molecules via strong Na+···O electrostatic
attractions. The nitrate ion completes the Na+
surroundings to an irregular octahedron. Perhaps the presence of
an alkaline metal ion partially stabilizes the strained
conformation of the ligand.