X-RAY ANALYSIS OF OXAZOLO[3,2-A]PYRIDINIUM CATION AND PRODUCTS OF ITS RING OPENING AND TRANSFORMATION
E. Babaev, V. Rybakov, S. Zhukov, A. Efimov
Chemistry Department, Moscow State University, 119899, Moscow, RUSSIA
E-mail: rybakov@biocryst.phys.msu.su
Keywords: X-ray, crystal
structure, oxazolo[3,2-a]pyridines, bonds alternation, ring
opening, recyclization, pyridones, indolizines,
pyrido[1,2-c]-1,2,4-triazines, oxazoles, butadienes,
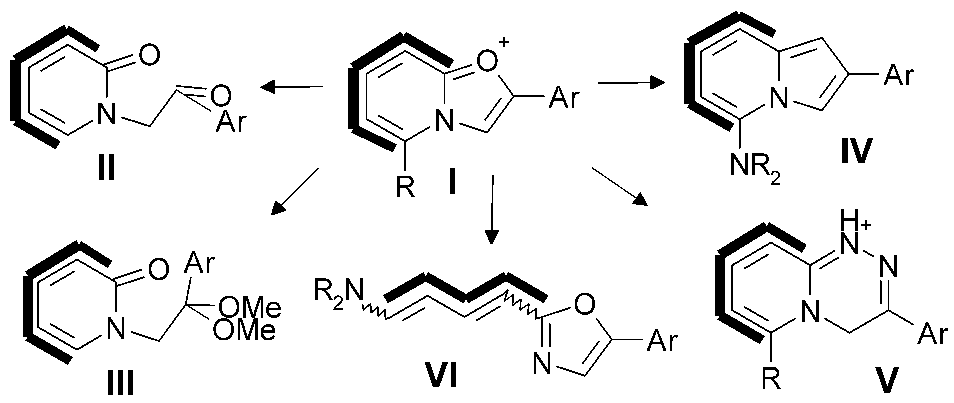
Recently we found several unusual ring opening
and ring transformation reactions of oxazolo[3,2-a]pyridinium
salts I. The structures of the initial heterocycle I
and various products of its reactions with nucleophiles (II
- VI) were investigated by X-ray method.

The structure of the initial cation (perchlorate I, R=H, Ar=Ph) displays clear alternation of bonds length in the pyridine fragment (shown by boldface line) [1]. This structural feature is "preserved" upon ring opening and transformation reactions in the products II - VI.
Pyridone II (obtained by keeping cation I
in aqueous HBF4) forms complex 3:1 with H3O+BF4-
[2]. In the case of ketal III (obtained by reaction of I
with MeONa [1]) the X-ray analysis confirmed the unique direction
of the oxazol ring cleavage. Family of previously unknown
5-aminoindolizines IV [3] was obtained by unusual
recyclization of 5-methyl derivatives of the cation I
(R=Me) under the action of secondary amines. In the structure of
these indolizines IVa (Ar=p-BrPh, NR2=morpholyl)
and VIb (Ar=p-NO2Ph, NR2=N(CH2)6)
the alternation of skeletal bonds is expressed at higher degree
than in any other known indolizines [4]. Therefore, the
conjugation of 5-amino group with the indolizine ring is
negligibly small. Compound V (R=Me, Ar=p-NO2Ph)
was obtained by reaction of I with N2H4.
NMR spectra of V in DMSO solution confirms the structure
of cation. However, according to X-ray data it is the complex of
two molecules of the free base with one molecule HClO4.
Reaction of secondary amines with cations I (R=H, Ar=Ph,
p-BrPh, p-NO2Ph, m-NO2Ph) lead to the
products of six-membered ring opening [1]. The configuration of
butadiene VI (NR2=morpholyl, Ar=p-NO2Ph)
previously assigned on the background of NMR data will be
discussed.
| Compound | I | II | III | IVa | IVb | V | VI |
| a, A | 5.762(1) | 14.020(2) | 8.812(2) | 7.578(1) | 6.614(1) | 14.560(4) | 7.058(4) |
| b, A | 14.469(3) | 14.020 | 10.026(1) | 9.371(2) | 15.005(1) | 14.618(9) | 10.698(4) |
| c, A | 16.598(4) | 33.005(5) | 15.152(2) | 11.632(5) | 8.821(1) | 14.579(5) | 42.00(2) |
| a, deg. | 112.53(2) | 90 | 90 | 86.11(2) | 90 | 90 | 90 |
| b, deg. | 83.35(2) | 90 | 95.32(2) | 88.69(2) | 102.94(2) | 114.69(3) | 90 |
| g, deg. | 90.29(2) | 120 | 90 | 70.03(1) | 90 | 90 | 90 |
| Z | 4 | 4 | 4 | 2 | 2 | 8 | 8 |
| Sp. gr. | P(-1) | R(-3) | P21/c | P(-1) | Pn | P21/c | Pb21a |