Aromaticity of Pyrazoles in Dependence on the Kind of Substitution
M. K. Cyranski1, T. M. Krygowski1, R. Anulewicz1, A. Puchala2 and D. Rasala2
1Department
of Chemistry, University of Warsaw, L. Pasteura 1, 02-093 Warsaw,
Poland
2Department of
Chemistry, Pedagogical University, Checinska 5, 25-020 Kielce,
Poland
Pyrazoles belong to a group of
p-electron systems with a strong
aromatic character. Aromaticity of nitrogen-containing p-electron
cyclic systems is sensitive to structural changes and
hence these systems have been widely used for studies of
multidimensionality of aromaticity. In the presentation crystal
and molecular structures of the
4-(4'-N,N-dimethylaminophenyl)-3,5-dimethyl-1,7-diphenyl-bis-pyrrazolo[3,4-b;4',3'-e]pyridine
and
3,5-dimethyl-1,4,7-triphenyl-bis-pyrrazolo[3,4-b;4',3'-e]pyridine
are reported, with R=0.0505 and 0.0538, respectively. Application
of the HOMA model [1, 2] to molecular geometry of the title
compounds enriched by the data for pyrazoles retrieved from
Cambridge Structural Database and from ab initio
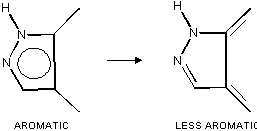
RHF/6-311G** calculations led to the conclusion formulated as a
rule that decrease of aromaticity of pyrazole is caused by an
increase of the double bond character of the exo-cyclic bonds
with substituents attached to C3 and C4 atoms in the ring. The
same is observed while calculating magnetic properties employing
NICS model [3].
