SYN-DIMERS AS NON-CENTROSYMMETRIC INTERMEDIATES IN THE SOLID-STATE SYNTHESIS OF CAGE DIMERS FROM 4-Aryl-1,4-Dihydropyridines
Andreas Hilgerothl, Ute Baumeister2, Günter Hempel3
1lnstitute of Pharmaceutical
Chemistry, Martin Luther University, Wolfgang-Langenbeck- Str.4,
06120 Halle, Germany,
E-mail: hilgeroth@pharmazie.uni-halle.de
2lnstitute of Physical Chemistry,
Martin Luther University, Mühlpforte 1, 06108 Halle, Germany
3lnstitute of Physics, Martin Luther
University, Friedemann-Bach-Platz 6, 06108 Halle, Germany
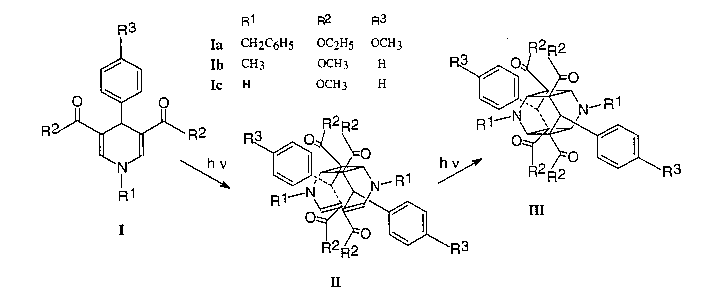
Crystalline 2,6-unsubstituted 4-aryl-1,4-dihydropyridines I have recently been reported to be highly light sensitive compounds forming either cage dimers III or anti-dimers, respectively [1]. The dimerization proved to be topochemically controlled by the shortest distance of potentially reacting double bonds [2,3]. With total yields of more than 90% for the formation of N substituted cage dimers they are of interest as attractive starting compounds for the synthesis of novel HIV-1 protease inhibitors [4].
A serie of derivates I with various substituents has been investigated by X-ray crystal structure analyses. The reacting molecules within these crystal packings are centrosymmetrically arranged. The cage formation may proceed via syn-dimers II, which could be isolated (yields: 40-70%), as was monitored by tlc. These syn-dimers cannot have a centrosymmetric molecular structure for constitutional reasons. One derivate IIa is characterized by X-ray crystal structure analysis. Furthermore, solid-state 13C-NMR investigations are reported which prove that the formation of III actually takes place via syn-dimers II by a primary [2+2]cycloaddition of a first pair of double bonds. This is followed by another [2+2]cycloaddition of the remaining pair of double bonds yielding centrosymmetric cage compounds III. Thus, centrosymmetry is broken in the first reaction and reestablished in the second one.
